 
The larger the molecule, the stronger the intermolecular forces become. This means that the diatomic molecules made up of two of the atoms will also become larger. This trend occurs because the melting and boiling points are determined by the strength of intermolecular forces of attraction acting between the halogen molecules.Īs you descend the group, the atoms become larger. When a halogen is heated up, it is the diatomic molecules which are separated from each other. Astatine is below iodine so has the highest melting and boiling points and must be a solid at room temperature.Įach of the halogens exist naturally as a pair of atoms covalently bonded together as diatomic molecules. Fluorine is above chlorine so would have the lowest melting and boiling points and must be a gas at room temperature. 
You can use this trend to predict the state of fluorine and astatine at room temperature. This is evidenced by the change in state at room temperature as you descend the group. You must also know the trends in physical properties of the halogens, focusing on chlorine, bromine and iodine.Īs you descend the group, the melting points and boiling points increase. Iodine – shiny dark purple/black solid crystals which turn into a purple vapour when heated.Bromine – red-brown liquid which turns into a red-brown vapour when heated.You must know the colours and physical states (at room temperature) of the halogens, focusing on chlorine, bromine and iodine. I'm guessing that's why you mentioned Socrates in your own comment.The halogens all have low melting and boiling points which means that they tend to be gases or liquids at room temperature. It just doesn't make sense that Wikipedia's definition could apply to a group 7A gas like Cl-it would be really weird if Cl by itself, elemental chlorine, underwent a chemical reaction, because it appears in nature as Cl 2, and I can't think of any possible explanation for this. But I don't think the Wikipedia definition matches the definition of reactivity in this context because the question seems to apply reactivity specifically to individual elements, while Wikipedia includes any chemical substance. Wikipedia defines reactivity as "the rate at which a chemical substance tends to undergo a chemical reaction," so this differs from my idea. So this question just got a whole lot more confusing.Īs for what OP meant by reactivity, my first thought was that reactivity just means how easily an atom reacts with another atom, which I believe is heavily influenced by how easily it is able to part from its valence electrons, since valence electrons are central to atomic bonding. Now that I'm thinking about it more carefully, I would imagine that the pattern of reactivity in group 7A would mimic that of group 2A, since the point I made about the shielding effect still applies to the halogens-that is, as in group 2A, each time you go down a row in group 7A, another layer of electrons is added, decreasing the effects of the nucleus's pull on the electrons. fit into the whole losing electrons idea. I'm not exactly sure anymore how F, Cl, Br, etc. I'm pretty sure OP meant group 7A, since it makes a lot more sense than would a seemingly arbitrary column of transition metals in the context of the question. A screenshot is preferable to a picture of your laptop screen. Please do not ask for help acquiring, preparing, or handling illicit substances or for help with any activity that does not fall within the confines of whatever laws apply to your particular location.īonus points: If submitting a picture please make sure that it is clear. Any infractions will be met with a temporary ban at the first instance and a permanent ban if there is another. It is also important that you describe the specific part of the problem you are struggling with. It is OK if you are a little (or a lot!) stuck, we just want to see that you have made an effort. Please complete any questions as much as you can before posting. 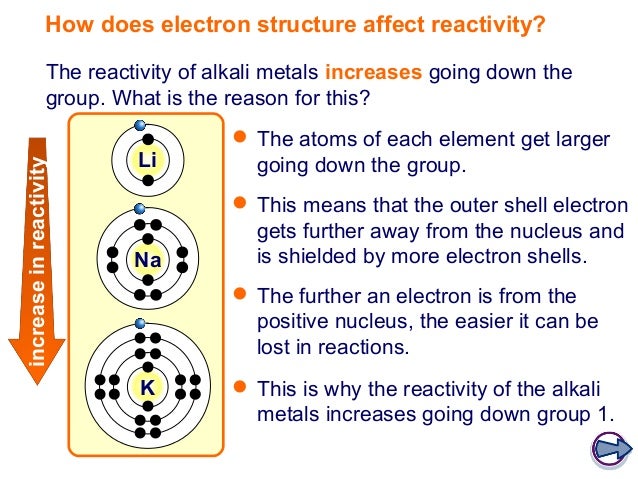
We will not do your homework for you, so don't ask. Please flair yourself and read over the rules below before posting. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
